- Blog
- Robert plutchik wheel of emotions
- Download mp3 music from youtube free
- Ancient bath glyph not there
- Omnisphere 2 crack no sound
- Farming simulator 15 xbox 360 lagging
- Pokemon gba text editor download emerald
- Graffiti letters alphabet wildstyl
- Join skype meeting on pc android
- Minecraft not installing windows 11
- Pdf memory bear sewing pattern free printable
- Nox app player for pc free download
- Hyde puff bar flavors
- Adobe premiere 2-0 serial number techspot
- Mobile home small log cabins texas
- Best family movies on netflix
- Download original spider solitaire for windows 10
- Google party planner
- Will installing windows 11 delete everything
- Sakura dungeon patch
- Arduino support package for matlab r2013a
- Free daily online crosswords
- Best free budget planner
- Genogram samples of 3 generation
- List of free wife porn sites
- Crock pot mac n cheese kraft
- Free font xeyewm din condensed bold
- Adobe after effects cs6 free serial key
- Noteburner apple music converter torrent
- Color palette picker wheel
- Rpg maker mv tileset d template
- Periodic table of elements molar masses
- Periodic table groups names chemistry
- Neon yellow among us logo
- Molar mass cl cl periodic table
- Mail forwarding forms usps
- Adobe after effect the piratebay
- Spectrum tv choice channels lineup
- Get rid of long bodied cellar spiders
- Minecraft 1-14-4 xray resource pack
- Half life 1 dev textures wad
- Free customized fantasy football draft cheat sheets
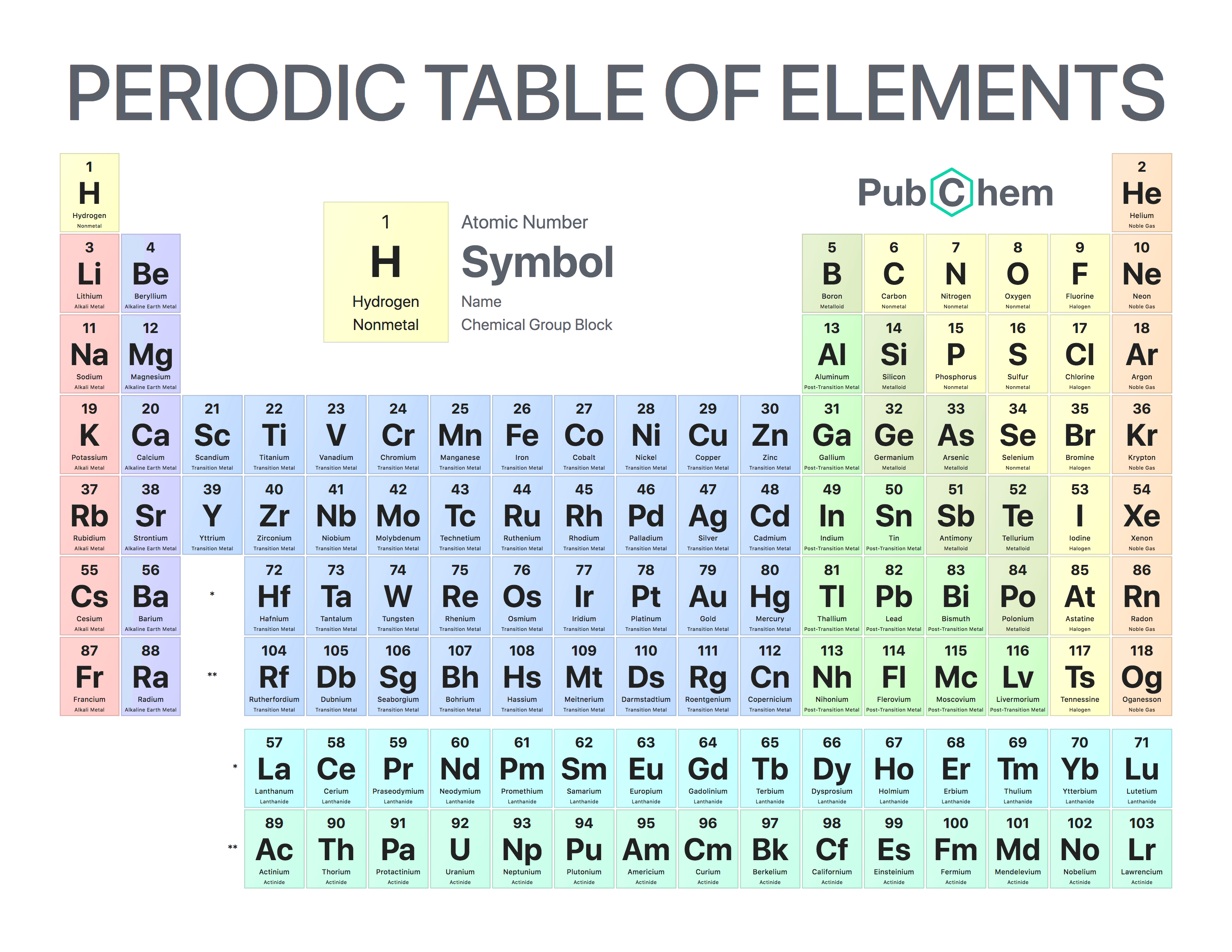
Elements that exhibit similar chemistry appear in vertical columns called groups (. The periodic table is used as a predictive tool that arranges of the elements in order of increasing atomic number. Nonmetals, in contrast, are generally poor conductors of heat and electricity and are not lustrous. 4.6: The Periodic Table of the Elements is shared under a CC BY 4.0 license and was authored, remixed, and/or curated by LibreTexts.

Of the metals, only mercury is a liquid at room temperature and pressure all the rest are solids. The vast majority of the known elements are metals. Metals-such as copper or gold-are good conductors of electricity and heat they can be pulled into wires because they are ductile they can be hammered or pressed into thin sheets or foils because they are malleable and most have a shiny appearance, so they are lustrous. The distinction between metals and nonmetals is one of the most fundamental in chemistry. Gold-colored lements that lie along the diagonal line exhibit properties intermediate between metals and nonmetals they are called semimetals. As expected, semimetals exhibit properties intermediate between metals and nonmetals.\) divides the elements into metals (in blue, below and to the left of the line) and nonmetals (in bronze, above and to the right of the line). Groups 1, 2, and 1318 are the main group elements, listed as A in older. The elements are stacked in such a way that elements with similar chemical properties form vertical columns, called groups, numbered from 1 to 18 (older periodic tables use a system based on roman numerals). One of the easiest ways to create a chemistry team name is to go back to the basics Create fun words using the periodic table of elements one of the first things you learn about as a budding scientist. Most solid nonmetals are brittle, so they break into small pieces when hit with a hammer or pulled into a wire. The rows are called periods, and they are numbered from 1 to 7. You may find inspiration in a famous chemist’s name or research. Nonmetals can be gases (such as chlorine), liquids (such as bromine), or solids (such as iodine) at room temperature and pressure. Nonmetals, in contrast, are generally poor conductors of heat and electricity and are not lustrous. The metals are on the bottom left in the periodic table, and the nonmetals are at the top right. 1: The Periodic Table Showing the Elements in Order of Increasing Z. In a neutral atom, the number of electrons equals the number of protons. Of the metals, only mercury is a liquid at room temperature and pressure all the rest are solids. The chemistry of each element is determined by its number of protons and electrons.

\) divides the elements into metals (in blue, below and to the left of the line) and nonmetals (in bronze, above and to the right of the line).

